![Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram](https://www.researchgate.net/publication/329439239/figure/fig3/AS:961808689008649@1606324469311/Calculated-isothermal-compressibility-kTdocumentclass12ptminimal.png)
Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram

The compressibility kappa of a substance is defined as the fractional change in volume of that substance for a given change in pressure : kappa = - 1V dVdP (a) Explain why

SOLVED: Isothermal compressibility, Kr (kappa defined as: Derive the formula that - van der waals gas would have for kappa Show the unit analysis on your answer in part and check if
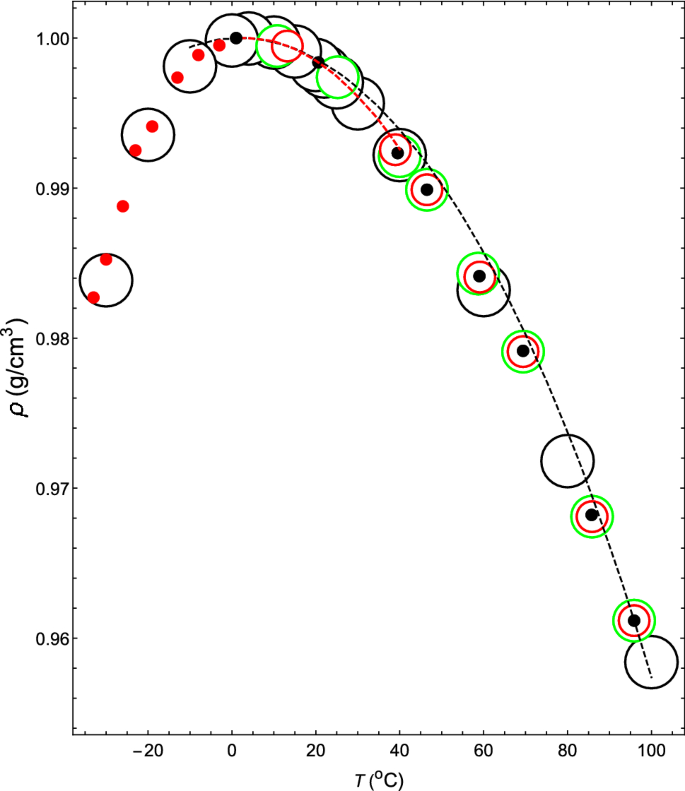
Thermodynamic mechanism of the density and compressibility anomalies of water in the range − 30 < T (°C) < 100 | Scientific Reports
![Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram](https://www.researchgate.net/publication/329439239/figure/fig3/AS:961808689008649@1606324469311/Calculated-isothermal-compressibility-kTdocumentclass12ptminimal_Q640.jpg)
Calculated isothermal compressibility κT\documentclass[12pt]{minimal}... | Download High-Resolution Scientific Diagram

Density, Speed of Sound, Compressibility and Related Excess Properties of Methane + n-Heptane at T = 303.15 K and p = 10 to 70 MPa | SpringerLink
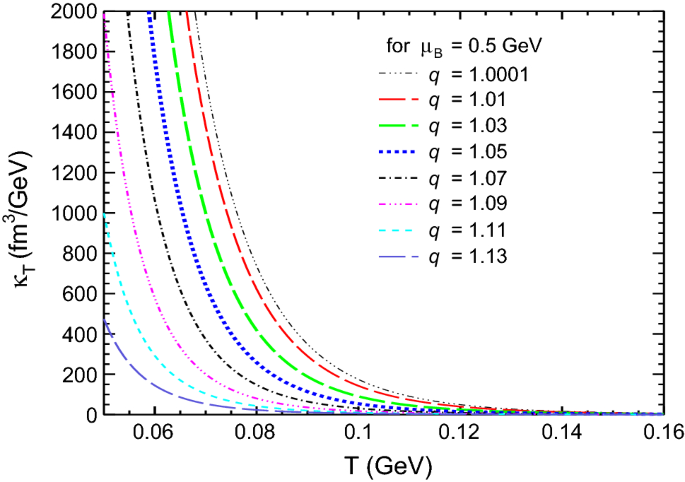
Dissipative properties and isothermal compressibility of hot and dense hadron gas using non-extensive statistics | SpringerLink

SOLVED:Generally, volume expansivity βand isothermal compressibility κdepend on T and P. Prove that: ((∂β)/(∂P))T=-((∂κ)/(∂T))P







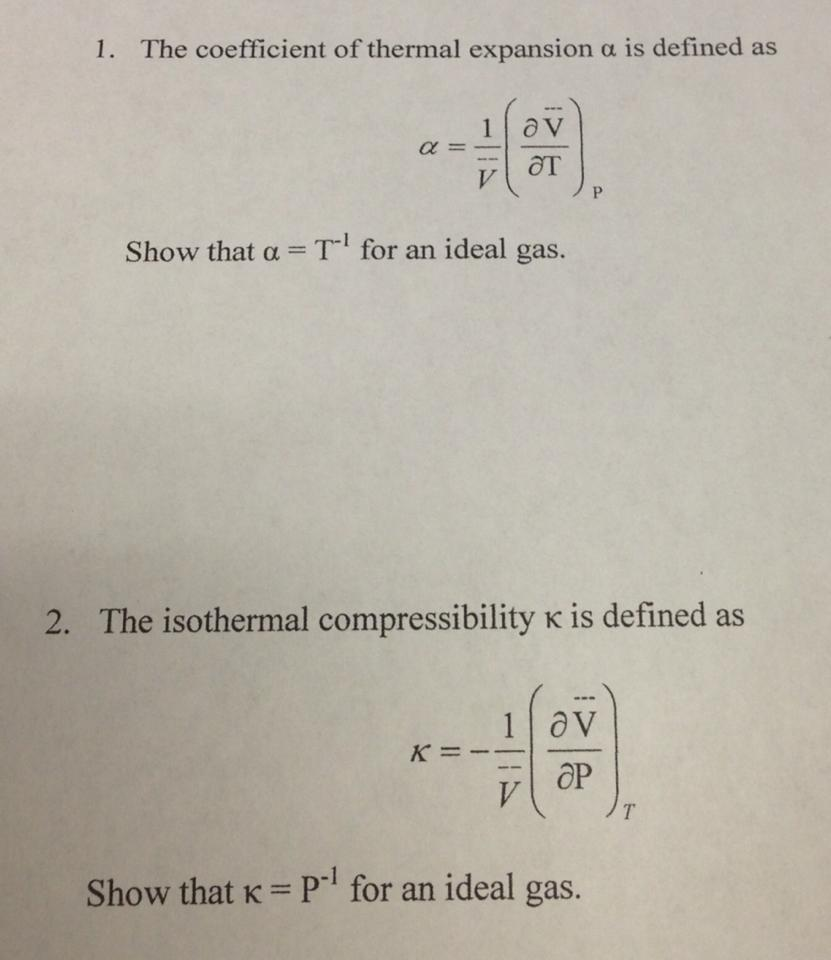

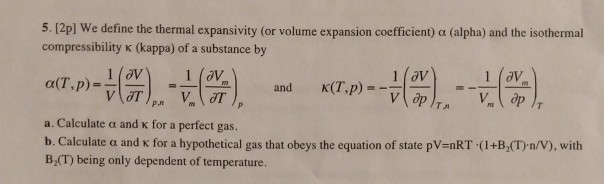


![Isothermal compressibility (κT\documentclass[12pt]{minimal}... | Download Scientific Diagram Isothermal compressibility (κT\documentclass[12pt]{minimal}... | Download Scientific Diagram](https://www.researchgate.net/publication/355714799/figure/fig8/AS:1084113842896899@1635484290073/Isothermal-compressibility-kTdocumentclass12ptminimal-usepackageamsmath.png)